Physics Words: Alpha scattering experiment
Simple Description
An experiment done in the early 20th Century that provided evidence for atoms being made of electrons orbiting a central, positive nucleus.
Further Detail
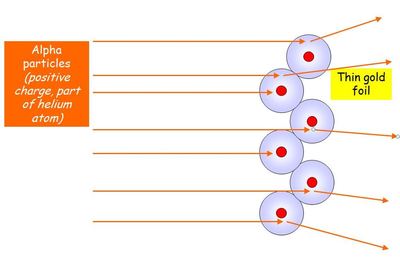
Rutherford proved the plum pudding model wrong by firing alpha particles straight at gold nucleii and showing that the alpha particles were deflected by varying angles. The Plum Pudding model couldn't explain this observation as it predicted all the alpha particles would travel straight through. Rutherford's experiment showed that:
1) Most of an atom is empty space (which is why most of the alpha particles went straight through)
2) Nucleii are extremely dense (which is why 1 in every 8,000 alpha particles were scattered by more than 90 degrees)
Related Words:
« Previous Word Next Word »
